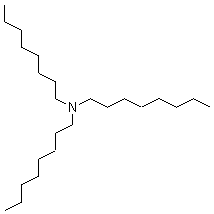
Is Trioctyl Tertiary Amine Hazardous?
TOA can be hazardous if not handled properly. It is toxic if swallowed, inhaled, or in contact with the skin. It may cause irritation or burns and can be harmful to aquatic life. Proper safety precautions, such as wearing protective clothing and gloves, should be followed when working with TOA.
Are There Any Alternatives to Trioctyl Tertiary Amine?
Depending on the specific application, there may be alternative compounds or methods available. In metal extraction processes, other amine-based extractants or different extraction techniques like solvent extraction or ion exchange may be used. It is recommended to consult with experts or professionals in the field for guidance on suitable alternatives.


 EN
EN
 jp
jp  ko
ko  fr
fr  de
de  es
es  it
it  ru
ru  pt
pt  ar
ar  tr
tr